You have no items in your shopping cart.
Cart summary

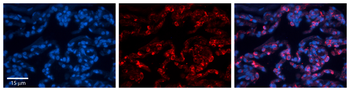
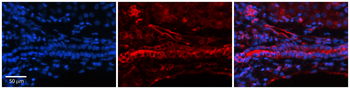
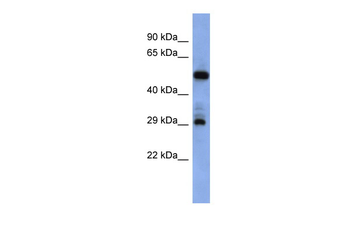
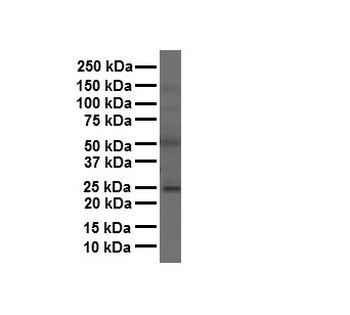
FOLR1 Antibody
Catalog Number: orb1906393
| Catalog Number | orb1906393 |
|---|---|
| Category | Antibodies |
| Description | FOLR1 Antibody |
| Species/Host | Human |
| Clonality | Monoclonal |
| Clone Number | Farletuzumab (MORAb-003; M3) |
| Tested applications | ELISA, FA, Functional Studies, IHC |
| Reactivity | Human, Primate |
| Isotype | IgG1 |
| Immunogen | The original parental mouse antibody was generated by immunizing (BALB/c X C57BL/6) F1 mice by intraperitoneal injection containing cultured LU-75(c) choriocarcinoma cells to produce hybridomas that generated the antibody LK26. Later on the Farletuzumab was generated by CDR grafting technique by taking CDRs of mouse parental clone LK26 and grafting them on human framework regions. |
| Concentration | 1 mg/ml |
| Conjugation | Unconjugated |
| Target | FOLR1 |
| UniProt ID | P15328 |
| Storage | Store at 4°C for up to 3 months. For longer storage, aliquot and store at -20°C. |
| Buffer/Preservatives | PBS with 0.02% Proclin 300. |
| Alternative names | FRA; Frα; Fr-α; FBP; LK26; Folate receptor 1; Fola Read more... |
| Note | For research use only |
| Application notes | This antibody was derived from the optimization of mouse parental LK26 antibody using a whole cell genetic evolution platform. This antibody possess growth-inhibitory activity on cells overexpressing FR-alpha. It elicited robust antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC) in vitro, and inhibited growth of human ovarian tumor xenografts in nude mice (PMID: 17346028). Immunohistochemistry studies determined that MORAb-003 infrequently stained the tubular epithelium, epithelium of the fallopian tube, and duct epithelium of the pancreas in both normal human and cynomolgus monkey tissues. Cynomolgus monkey was therefore considered an appropriate toxicology model (PMID: 17346028). This antibody was also found to antagonize the activity of human FRα, resulting in the loss of growth advantage conferred by overexpression of FRα under conditions which bracket physiological (10–100 nM) folate concentrations (Routhier et al, 2006). A phase I study in patients with platinum resistant ovarian cancer revealed that MORAb-003 was well tolerated in patients with epithelial ovarian cancers and may may have activity in platinum-resistant patients (Konner et al, 2006; Bell-McGuinn et al, 2007). In a phase 2 trial in patients with platinum-sensitive ovarian cancer, patients received single-agent farletuzumab or farletuzumab combined with carboplatin and paclitaxel (or docetaxel), followed by farletuzumab maintenance until progression. Of the 47 patients who received farletuzumab, 80.9% had normalization of their CA125, and a complete or partial objective response rate was achieved in 75% with combination therapy (Herzog et al, 2016). It was also reported that farletuzumab with carboplatin and taxane may enhance the response rate and duration of response in platinum-sensitive ovarian cancer patients with first relapse after remission of 6–18 months (PMID: 23474348). |
| Expiration Date | 12 months from date of receipt. |
FOLR1 antibody [orb578055]
IHC, WB
Bovine, Canine, Equine, Guinea pig, Mouse, Rabbit, Rat
Human
Rabbit
Polyclonal
Unconjugated
100 μlAnti-FOLR1 Reference Antibody [orb1806347]
ELISA, FA, FACS, Kinetics
Human
Monoclonal
Unconjugated
50 μg, 100 μg, 1 mg, 5 mgAnti-FOLR1 Reference Antibody [orb1806348]
ELISA, FA, FACS, Kinetics
Human
Monoclonal
Unconjugated
50 μg, 100 μg, 1 mg, 5 mgFOLR1 antibody [orb213943]
IF, IH, WB
Human, Mouse, Primate, Rat
Rabbit
Polyclonal
Unconjugated
200 μl, 100 μl, 30 μl
Submit a review
Filter by Rating
- 5 stars
- 4 stars
- 3 stars
- 2 stars
- 1 stars